Vayu - Medical Refrigerator Monitoring Data Logger
The Vayu - Medical Refrigerator Monitoring Data Logger is a mission-critical scientific instrument designed to eliminate "thermal blind spots" in clinical and pharmaceutical storage environments. At Vega Calibration and Validation Services LLP, we recognize that the integrity of life-saving vaccines, reagents, and biological samples hinges on absolute thermal stability. Vayu provides a 24/7 digital "thermal biography" of your assets, streaming laboratory-grade data over global 4G/GSM networks directly to a secure, 21 CFR Part 11 compliant cloud. For specialized research and ultra-low temperature requirements—such as -80°C freezers or liquid nitrogen dewars—a Greater Range External Probe version is available, supporting measurements from -200°C to +250°C. Whether you are managing a central pharmacy in Mumbai, a research lab in Hyderabad, or a diagnostic chain in Dubai, Vayu ensures your environmental parameters remain in a "validated state," providing the technical precision and audit-readiness required by modern Quality Managers.
The VAYU is an advanced multi-use tracking device designed for demanding industrial applications such as cold chain logistics, pharmaceuticals, and food transportation.
It features an IP64-rated enclosure, providing protection against dust and water splashes, making it suitable for harsh operating environments.
The device operates effectively within a temperature range of –20°C to 60°C, ensuring reliable monitoring in both refrigerated and ambient conditions.
It is powered by a 3.7V / 4000mAh lithium battery, offering a long operational life of up to 60 days at a default 1-hour recording interval.
The logger supports 4G and 2G communication technologies (LTE CAT1 and GSM), enabling stable and uninterrupted global data transmission.
It delivers high measurement accuracy, with ±0.3°C for temperature and ±3% relative humidity, ensuring precise environmental monitoring.
The system includes a large internal storage capacity of up to 17,000 records, preventing data loss during temporary network interruptions.
It integrates multiple positioning technologies including GPS, WiFi, and LBS, allowing accurate real-time location tracking of shipments.
Elaborate Product Offerings
Specialized Product Capabilities
The device includes advanced motion sensors that detect and record vibrations during transportation, helping identify improper handling or shock events.
A light detection sensor is integrated to monitor low-light conditions and detect exposure to external light sources.
The system can identify door opening and closing events or switching box activities, helping monitor unauthorized access or handling during transit.
All collected data is processed through the VEGA Equipment Management System, which acts as the central platform for device monitoring and data management.
The platform provides comprehensive data processing features that help users analyze environmental conditions and device performance effectively.
It offers FDA-compliant statistical reporting, supporting regulatory documentation and audit readiness.
The system enables trajectory analysis, allowing users to track shipment routes and understand environmental conditions throughout the logistics journey.
Instant real-time alarms and notifications are generated whenever predefined thresholds are exceeded, enabling immediate corrective actions to protect valuable assets.
Technical Specifications


Get in touch
Share with visitors how they can contact you and encourage them to ask any questions they may have.
Phone
123-123-1234
email@email.com












FCC
EN 12830
US FDA 21 CRF PART 11
WHO-GMP
RoHS
CE
Regulatory Compliance & Certifications


ISO/IEC 17025:2017 NABL Accredited Calibration Laboratory
Vega operates a NABL-accredited calibration laboratory and holds a valid certification in accordance with ISO/IEC 17025:2017, demonstrating our commitment to maintaining the highest standards in calibration and testing. This accreditation confirms our technical competence, ensuring that all calibration processes are performed with precision, reliability, and adherence to internationally recognized standards.
Key Benefits
Ensures metrological traceability and high measurement accuracy.
Demonstrates the technical competence and reliability of our calibration processes.
Provides international recognition and credibility for calibration and test results.
Supports regulatory compliance and quality assurance in pharmaceutical, healthcare, food, and other regulated industries.
User-Friendly Dashboards for Complete Operational Visibility
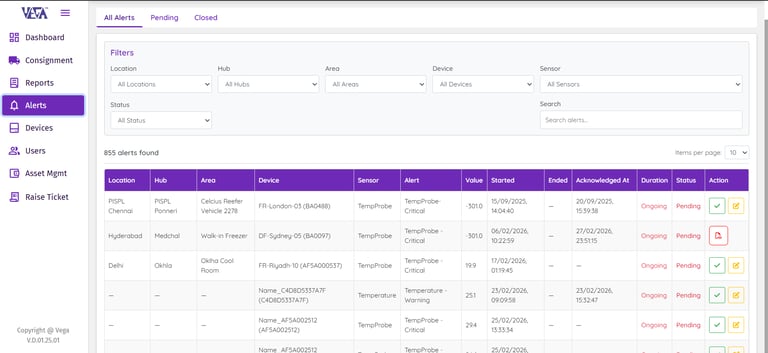
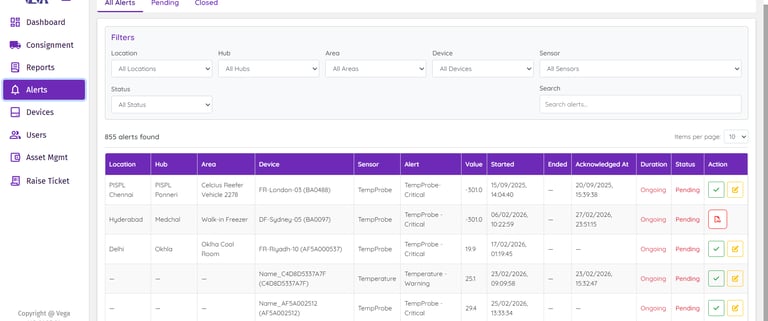
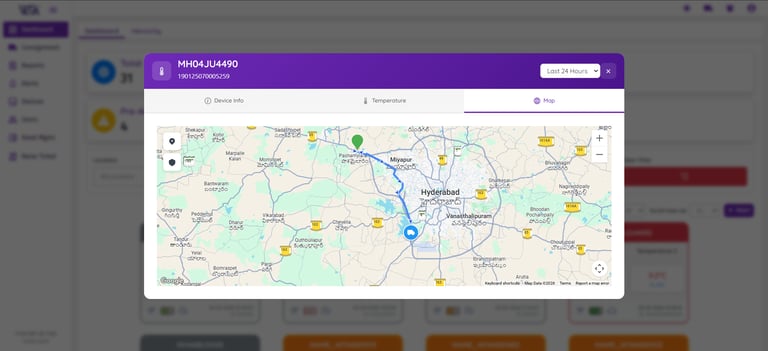
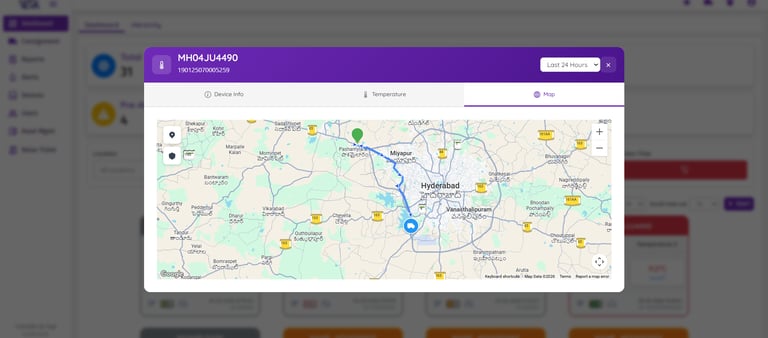
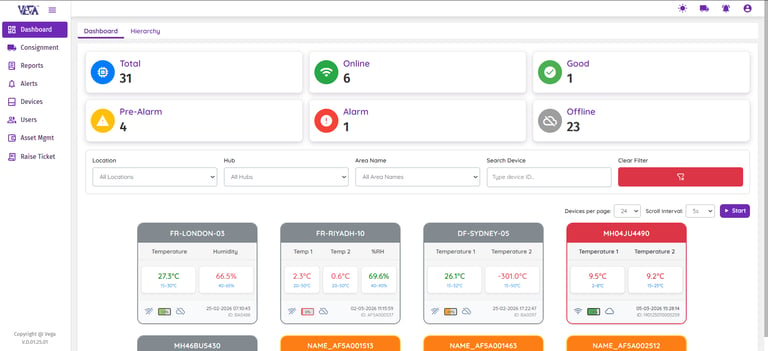
Data Logger Status Overview
Get an instant, real-time snapshot of your entire fleet of data loggers. Monitor device status, battery health, and critical alerts from a single dashboard. This enables continuous system operation while allowing teams to identify potential issues early.
Real-Time Alerts & Analytics
Visualize essential temperature and humidity data through customizable charts and graphs. Easily track trends, detect deviations, and perform detailed analysis to help maintain optimal environmental conditions for sensitive and temperature-critical goods.
Real-Time Alerts for Your Devices
Receive instant alerts from your devices to stay informed about any issues as they occur. This enables you to quickly identify shortcomings, take immediate corrective action, reduce downtime, and protect valuable time and products.
Industry Segments we Serve
Global Bio-Pharmaceutical and Vaccine Manufacturing
In the massive pharmaceutical clusters of Ahmedabad, Hyderabad, and Baddi, the Vayu medical refrigerator monitoring data logger is an indispensable tool for maintaining the "validated state" of raw materials and finished drug batches. This segment handles life-saving vaccines, biological reagents, and high-value APIs that are extremely sensitive to thermal fluctuations; even a subtle drift during a stability test or within a sterile cold room can render a multi-million dollar batch ineffective or legally non-compliant. Our loggers provide the continuous, high-resolution data required to satisfy the US FDA's 21 CFR Part 11 and EU GMP Annex 11 requirements. By utilizing Vayu, manufacturers ensure that every stage of the production cycle is protected by a documented and secure digital record. This level of technical oversight is essential for surviving inspections from global health authorities and maintaining the integrity of products destined for international markets like Jeddah or Bahrain. The monitors ensure that data integrity is backed by scientifically sound, unalterable digital records that prove the environment remained within validated limits at all times. The ability to receive an active alert during a power failure provides a critical window for intervention, safeguarding both the chemical assets and the corporate reputation.
Clinical Research Organizations (CROs) and Bio-Banking
The biotechnology research corridors of Bangalore, Jaipur, and Goa utilize the Vayu logger to protect invaluable biological samples, serum, and experimental drug batches stored in laboratory refrigerators. In these facilities, thousands of samples are archived for years, where the slightest thermal deviation can lead to irreplaceable research loss and the invalidation of clinical trial results. Our monitoring systems provide the high-frequency data stream and real-time alerts needed to ensure these environments remain within their narrow tolerance bands in cities like Chennai and Dubai. The Greater Range External Probe version is frequently used to monitor the interior of liquid nitrogen tanks or ultra-low freezers while the logger maintains a cellular connection outside. By maintaining a scientific, tamper-proof record of the storage environment via the cloud, CROs can significantly improve data reliability and satisfy the rigorous standards required for global medical trials. This data-driven approach upholds the reputation of Indian technical excellence in the global market through unalterable digital evidence and real-time environmental security that synchronizes directly with international scientific quality standards, ensuring that every sample's history is fully documented and audit-ready.
Diagnostic Laboratories and Large-Scale Healthcare Chains
Large diagnostic chains with centers in Lucknow, Jaipur, and Kolkata face the challenge of maintaining consistent environmental logs for reagents and patient samples across hundreds of decentralized locations. By implementing a standardized use of Vayu medical loggers, the corporate Quality Manager in Mumbai can oversee every regional site via a single customized dashboard. The system automatically flags any refrigerator that has experienced a temperature excursion, whether in the humid heat of Indore or the arid climate of Riyadh. This use case transforms compliance from a localized, manual task into a streamlined, automated corporate function. It ensures that every branch is adhering to the same high standards, protecting diagnostic accuracy and simplifying the process for international audits. The high-resolution data provided by the loggers allows for better trend analysis and predictive maintenance of the laboratory equipment, reducing the total cost of quality and ensuring all global branches meet rigorous benchmarks for environmental security without manual intervention.
Specialized Chemical and High-Tech Material Research
The high-tech research hubs of Noida, Manesar, and Pune utilize the Vayu logger to protect specialized chemical reagents and sensitive materials during critical stability aging studies. In these industries, even minor temperature fluctuations can cause chemical degradation or physical stress on experimental materials. Furthermore, high-value assets often require monitoring in extreme temperatures; the Greater Range External Probe version is used to monitor high-temperature ovens or cryogenic chambers. By maintaining a scientific, tamper-proof record of the research environment via the cloud, these firms can significantly reduce defect rates and ensure their results meet the rigorous standards required for global partners. This data-driven approach upholds the reputation of Indian technical excellence in the global market through unalterable digital evidence and real-time environmental security. The ability to receive an active alert during a power failure or equipment malfunction provides a critical window for intervention, safeguarding expensive research assets and ensuring that experimental results are always scientifically defensible.
Industrial Use Cases
Real-Time Safeguarding of Ultra-Low Temperature (ULT) Samples
One of the most vital use cases involves the storage of priceless biological samples in -80°C freezers in labs across Delhi or Mumbai. These samples are extremely sensitive; a single failure in the cooling system can lead to total research destruction within hours. By deploying the Greater Range Probe version of Vayu, the lab team receives an immediate SMS alert the moment the temperature rises above a critical threshold. If a compressor fails in a facility in Dubai or if a door is left open at a lab in Ahmedabad, Vayu’s GSM connectivity ensures the excursion is flagged instantly. This allows for proactive intervention—such as moving the stock to a backup freezer—preventing the loss of decades of scientific work. This use case is a cornerstone of Good Laboratory Practices, providing a documented "thermal biography" for every rack. It ensures that health authorities in Saudi Arabia and Bahrain have the live data needed to verify the integrity of the research, ensuring national health security through unalterable, system-integrated digital documentation.
Validation and Real-Time Mapping of Pharmaceutical Storage Units
Before a new large-scale refrigerator or cold room in Noida, Hyderabad, or Pune can be commissioned for GxP-compliant storage, it must undergo a rigorous "Thermal Performance Validation" study. Vayu loggers are placed in "worst-case" positions to identify how the cooling system performs under extreme ambient heat. Unlike traditional loggers where data is checked at the end of the study, Vayu allows Quality Managers to view the mapping progress live on a cloud dashboard. This mapping is a mandatory requirement for WHO and GMP compliance. Once the mapping is complete, the loggers provide the data needed to create a final validation report. This use case transforms environmental data into operational intelligence, proving to auditors that the equipment is fit for purpose and capable of maintaining uniformity even under maximum loading conditions. By analyzing these live data points, managers can optimize set-points in real-time, reducing energy costs while maintaining 100% safety for the products inside, ensuring the infrastructure is functioning correctly.
Centralized Multi-Site Compliance for Regional Diagnostic Networks
Large diagnostic companies with labs across Jaipur, Kolkata, and Chandigarh face the difficulty of maintaining consistent logs for thousands of reagent refrigerators. By implementing Vayu loggers, the corporate Quality Manager in Mumbai can oversee every regional site via a single customized dashboard. The system automatically flags any refrigerator that has experienced a temperature excursion, perhaps due to a local power outage in Lucknow or a door-seal failure in Coimbatore. This use case transforms compliance from a manual, error-prone task into a streamlined, automated corporate function. It ensures that every branch is adhering to the same high standards, protecting reagent efficacy and simplifying the process for international audits. The high-resolution data provided by the loggers allows for better trend analysis and predictive maintenance of the lab infrastructure, reducing the total cost of quality and ensuring all global branches meet rigorous benchmarks for environmental security without manual intervention, saving thousands of man-hours in data collection and proving absolute care to auditors.
Cryogenic Monitoring and Security for Stem Cell Research
Irreplaceable stem cell lines and germplasm stored in liquid nitrogen in research labs in Jaipur and Kolkata require incredibly stable environments to prevent genetic drift or contamination. The Greater Range Probe version of Vayu is used to provide continuous, live tracking of the nitrogen dewar micro-climate. Because these biological assets are irreplaceable, the monitoring system acts as an objective, live witness to the environmental stability inside the tank. The automated, real-time reports are used to prove to international research partners in Dubai or Bahrain that the cryogenic process was capable of maintaining the rigorous conditions required for medical research. This use case highlights the system's role in genetic preservation, where the technical precision of the logger data is the only way to ensure that medicinal strains are protected for future generations. Vayu provides a secure and transparent environmental record that satisfies the most demanding global research and insurance standards, ensuring the history of medicinal discovery is preserved through scientifically sound and instantaneous documentation.
Compliance Requirements
The VAYU is designed to support compliance with major international audit and regulatory standards.
It aligns with FDA 21 CFR Part 11 requirements by providing secure, tamper-proof, and unalterable electronic data records.
The system generates automated analysis reports and detailed statistical data, ensuring reliable documentation and easy regulatory verification.
It ensures complete quality traceability, allowing organizations to track and verify environmental conditions throughout the monitoring process.
The VEGA Cloud Platform produces robust and systematic digital records that support regulatory inspections and internal quality audits.
The system supports compliance with Good Manufacturing Practices (GMP), ensuring proper monitoring and documentation in manufacturing environments.
The platform provides time-stamped monitoring records, offering verifiable proof of environmental consistency during routine inspections and compliance checks.
The device’s high measurement accuracy supports organizations following **ISO/IEC 17025:2017 standards for testing and calibration laboratories.
Why Vega is Your Ideal Partner
Partnering with Vega Calibration And Validation Services LLP ensures reliable, on-time support and advanced IoT monitoring solutions regardless of geographical location.
The VAYU can be rapidly deployed across major industrial hubs in India, including Mumbai, Delhi, Bangalore, Hyderabad, Ahmedabad, and Kolkata.
Vega also supports operations in key manufacturing and pharmaceutical regions such as Indore, Chandigarh, and Manesar, ensuring consistent monitoring and technical assistance.
Dedicated operational teams provide continuous support across important logistics hubs including Pune, Noida, Jaipur, Panvel, and Navi Mumbai to maintain uninterrupted cold chain operations.
Vega also supports southern and international markets, including Chennai, Kochi, Coimbatore, and deployments across Saudi Arabia, including Riyadh, Jeddah, and Dammam, ensuring global reliability and technical support.
Frequently Asked Questions
Q: What is the battery life of the VAYU data logger?
A: The VAYU system features a built-in rechargeable 3.7V/4000mAh Lithium battery that lasts up to 60 days based on a default 1-hour reporting interval.
Q: What is the benefit of the external probe version for live tracking?
A: The external probe allows for monitoring extreme temperatures (-200°C to +250°C) inside a container or specialized shipper while keeping the main unit outside for signal strength. This is critical for ultra-low temperature freezers or industrial ovens in Pune and Hyderabad.
Q: Is the VEGA cloud platform accessible on mobile devices?
A: Yes, the VEGA Cloud Platform is fully mobile and desktop responsive, offering real-time alerts, map reports, and scheduling features on any smart device.
Q: Can I set different alert levels for different team members?
A: Yes. The Vayu software allows for sophisticated escalation matrixes. For example, a minor deviation can send an email to the facility manager in Riyadh, while a critical temperature breach can trigger an urgent SMS to the Quality Head in Mumbai, ensuring the right people are notified at the right time.
Q: Can I set up different alert thresholds for different types of medical equipment?
A: Yes. The Vayu software is highly flexible. You can configure unique alarm profiles for different assets—such as 2°C to 8°C for reagent fridges or -75°C to -85°C for ULT freezers—ensuring that Quality Managers in Riyadh or Mumbai receive the correct alerts for their specific samples.
Customer Testimonials
"Vega’s Vayu medical loggers have completely changed our cold chain risk management in Hyderabad. Receiving an instant SMS the moment a stability chamber deviates in Dubai gives our quality team absolute peace of mind." — Quality Director, Major Pharma Exporter.
"For our bio-bank in Noida, the external probe version of Vayu is flawless. We can monitor our ultra-low freezers and liquid nitrogen tanks live from our head office in Mumbai and receive alerts before any sample is at risk." — Principal Scientist, Biotechnology Research Firm.
"We use Vayu for our multi-site diagnostic network across Indore, Pune, and Jaipur. The live cloud dashboard and instant excursion alerts ensure we are 100% compliant with global GDP standards." — Operations Head, International Diagnostic Chain.
Support
Contact
support@vegabazaar.com
+91-76-96-39-63-93
© 2025. All rights reserved @ Vega Enterprises
Head Office
A202A, Jaswanti Allied Business Centre Kachpada, Ramchandra Ln, Extn, Malad West, Mumbai, Maharashtra 400064